
The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model. Electrons can be arranged correctly through orbits from elements 1 to 18. Therefore, the order of the number of electrons in each shell of the radon(Rn) atom is 2, 8, 18, 32, 18, 8. Therefore, a radon atom will have two electrons in the first shell, eight in the 2nd orbit, eighteen electrons in the 3rd shell, and thirty-two in the 4th shell.Īccording to Bohr’s formula, the fifth shell will have twenty-six electrons but the fifth shell of radon will have eighteen electrons and the remaining eight electrons will be in the sixth shell. 
That is, the number of electrons in radon is eighty-six. The atomic number is the number of electrons in that element. Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons. The maximum electron holding capacity in N orbit is 2n 2 = 2 × 4 2 = 32. 
The maximum electron holding capacity in M orbit is 2n 2 = 2 × 3 2 = 18. The maximum electron holding capacity in L orbit is 2n 2 = 2 × 2 2 = 8. The maximum electron holding capacity in K orbit is 2n 2 = 2 × 1 2 = 2. The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. Scientist Niels Bohr was the first to give an idea of the atom’s orbit. Radon atom electron configuration through orbit For example Aufbau principle, Hund’s principle, and Pauli’s exclusion principle. Coenzyme B12 provides the C-Co bond, which is part of the reactions.Radon atom electron configuration (Bohr model)Įlectron configuration through orbitals follows different principles. The absence or low presence of cobalt can cause anemia.Ĭobalamin proteins use the corrin ring to hold the cobalt unit. It makes up cobalamin, also called vitamin B12. Biological function of cobaltĬobalt is needed in all animals and in humans as well. In the absence of nuclear war, the risk arises from improper maintenance or handling of radiotherapy units. This is why certain nuclear weapons called dirty bombs are created. If a nuclear confrontation were to occur, the emission of neutrons could convert iron into Co-60, greatly increasing the consequences of radioactive contamination.Ingestion of Co-60 can cause a certain amount to accumulate in the tissues, which is eliminated slowly. Co-60 is radioactive, exposure to its radiation can cause cancer.Generally, cobalt compounds should be handled with care as the metal has mild toxicity. Finely separated metallic cobalt powder is flammable.For this reason, these fonts have ceased to be popular in the West. The Cobalt-60 source is composed of a useful life of about 5 years, but when this duration is exceeded, the isotope is still quite radioactive. Metal normally develops a fairly fine dust which makes radiation shielding difficult. The source used has a radius of 2 cm and produces the presence of penumbral parts that diffuse the radiation that surrounds its direction. it is therefore difficult to be able to encapsulate it to avoid radioactive contamination.Ĭobalt-60 generates two gamma rays that have energies of 1.33 MeV and 1.17 Mev. Isotopes lighter than the stable Co-59 are primarily decayed by electron capture creating iron isotopes, while those heavier than the stable isotope are decayed by beta emission giving rise to nickel isotopes.Ĭobalt-60 is used in radiotherapy instead of radium because of its economic price and also because radium decays into radon and it is a radioactive element in the form of a gas. 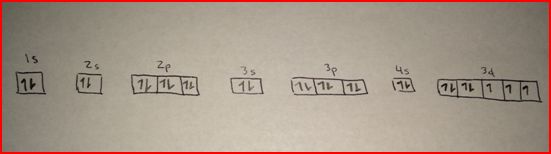
The atomic mass of isotopes of this element is between 50 amu and 73 amu. Cobalt also has four meta-states, each of which has half-lives of less than 15 minutes. The other radioactive isotopes have half-lives of less than 18 hours and almost all of them less than one second. The most stable are Co-56, Co-57 and Co-60 whose half-lives are 70.86 days, 271.79 days and 5.2714 years respectively. The boiling point of this element is 2927.85 degrees Celsius or 3200 Kelvin and the melting point is 1495.85 degrees Celsius or 1768 Kelvin. This chemical element has a gray tint, is metallic and belongs to the group of transition metals, its atomic number is 27 and it is represented by the symbol Co. The covalent radius of cobalt is 126 pm, the Bohr radius or atomic radius is 152 pm, while the average radius is 135 pm.Ĭobalt is solid in its natural form, specifically a ferromagnetic solid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
